LD-50 Lab Report:
Collaborators: Christina Fillipini, Julianne Rodgers, Logan Shytle, and collaborated with the entire class to compile the data
Introduction:
The LD-5o lab tests the effects of salt on the germination and growth of plants. By definition, LD-50 is the amount of a material given all at once, which causes death of 50% of a test population. To see these results a dose-response curve, which is a graphical representation of the relationships between the does of a stressor and the biological response, is used. In this lab serial dilutions are used to stepwise dilute substance into solution with constant dilution. In other words serial dilutions are used to "dilute the dilution." The purpose of this lab is to determine the LD-50 of salt for radish seeds and to determine how salt affects the initial growth of radish plants. It is important to know how salt affects plants because salt is commonly used on roads before and during ice and snow storms. "Salting roads works by altering the freezing point of water. Water with a higher salt content has a lower freezing point than water with less salinity...Though salt can be effective, it does have some negative impacts on the environment" (Tuthill). Knowing how salt affects the plants, specifically the LD-50, will help to discover what can be done to reduce the negative impacts salting roads has on the environment.
Hypothesis:
If the concentration of 18 g/L of salt is used on the radish seeds than half of the radish seeds will die because the radish seed will not be able to survive with high amounts of salt.
Parts of the Experiment:
- Independent Variable: The amount of salt in each
concentration
- Dependent Variable: The germination of the seeds
and the growth of the root
- Control Variables: The materials, the amount of
seeds in each different concentration, the amount of sunlight, the amount of
time that the seeds were given to germinate
- Experimental Group: The seeds in the different
concentrations of salt
- Control Group: The group of seeds in 0% salt.
Materials:
-6 test tubes
-test tube rack
-60 radish seeds, 10 per sample
-6 paper towels
-6 zip-lock bags
-pipette
-distilled water
-concentrated salt solution (18.0 grams/ liter of water)
-ruler
-test tube rack
-60 radish seeds, 10 per sample
-6 paper towels
-6 zip-lock bags
-pipette
-distilled water
-concentrated salt solution (18.0 grams/ liter of water)
-ruler
Methods:
Part 1: Serial Dilution
1. Set up 6 test tubes on a test rack
2. Begin by pouring 20ml of the salt solution (18 g/L) into the first test tube.
2. Then pour 10ml of solution into test tube 2
3. Add 10ml of distilled water (there should now be 20ml of mixture in test tube 2)
4. Then pour 10ml of solution from test tube 2 into test tube 3.
5. Add 10ml of distilled water (there should now be 20ml of mixture in test tube 3)
6. Pour 10ml of solution from test tube 3 into test tube 4.
7. Add 10ml of distilled water (there should now be 20ml of mixture in test tube 4)
8. Pour 10ml of solution from test tube 4 into test tube 5
9. Add 10ml of distilled water (there should now be 20ml of mixture in test tube 5)
10. Pour 10ml of distilled water into test tube 6. This is the control.
Part 2:
1. Label the six plastic bags with the 6 different concentrations (100%, 50%, 25%, ect.)
2. Begin by wetting (not soaking) one of the paper towels with the 100% concentration.
3. Place the 10 seeds onto the paper towel, fold it, and put the paper towel into the plastic bag, sealing the bag.
4. Repeat steps 2-3 for all the different concentrations.
5. Put bags in a cool dry place for a few days
6. Check seeds in a few days to see if more water needs to be added
Part 3: Collecting Data
1. Remove the seeds from one bag and count the number germinated, record the percent.
2. Repeat step one for each different concentration.
3. Measure the length of the radicle for the germinated seeds in mm, record.
4. Take the average of the radicle lengths of the germinated seeds for each different concentration, record.
1. Set up 6 test tubes on a test rack
2. Begin by pouring 20ml of the salt solution (18 g/L) into the first test tube.
2. Then pour 10ml of solution into test tube 2
3. Add 10ml of distilled water (there should now be 20ml of mixture in test tube 2)
4. Then pour 10ml of solution from test tube 2 into test tube 3.
5. Add 10ml of distilled water (there should now be 20ml of mixture in test tube 3)
6. Pour 10ml of solution from test tube 3 into test tube 4.
7. Add 10ml of distilled water (there should now be 20ml of mixture in test tube 4)
8. Pour 10ml of solution from test tube 4 into test tube 5
9. Add 10ml of distilled water (there should now be 20ml of mixture in test tube 5)
10. Pour 10ml of distilled water into test tube 6. This is the control.
Part 2:
1. Label the six plastic bags with the 6 different concentrations (100%, 50%, 25%, ect.)
2. Begin by wetting (not soaking) one of the paper towels with the 100% concentration.
3. Place the 10 seeds onto the paper towel, fold it, and put the paper towel into the plastic bag, sealing the bag.
4. Repeat steps 2-3 for all the different concentrations.
5. Put bags in a cool dry place for a few days
6. Check seeds in a few days to see if more water needs to be added
Part 3: Collecting Data
1. Remove the seeds from one bag and count the number germinated, record the percent.
2. Repeat step one for each different concentration.
3. Measure the length of the radicle for the germinated seeds in mm, record.
4. Take the average of the radicle lengths of the germinated seeds for each different concentration, record.
Data & Data AnalysisData Analysis:After the LD-50 lab was completed and the data was compiled, some trends within the data can be seen. As the amount of salt in the solution increased, the average radicle length of the seeds decreased. Seeds that were in the concentration of 6.25% salt solution had radicles that averaged around 29.496mm, while seed that were in the concentration of 100% salt solution had radicles that averaged around 4.739. This data shows that salt decreases the radical length in the germinated seeds. However, the data does not show as clear a correlation between the salt concentration and the average percent of seeds germinated. It is clear from the table that the smallest percent, only 74%, of seeds germinated in the 100% salt solution. Whereas it is unclear as to why only 84% of radish seeds germinated in the 25% concentration while 95.7% germinated in both the 50% and 6.25% concentrations.
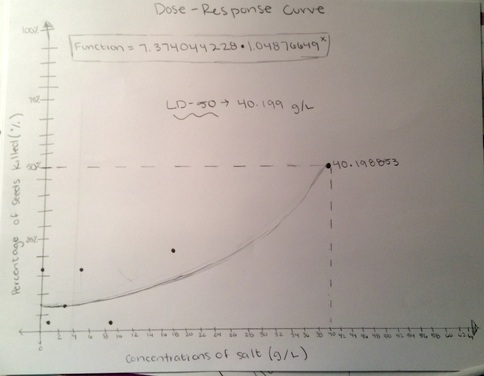
When the percentage of the seeds killed (seeds not germinated) is plotted based on the concentrations of salt per g/L a dose-response curve is formed. The general function used to be able to see the dose-response curve and ultimately find the LD-50 was: (7.374044228)x(1.04876649^x). This function is an estimate of the line of best fit, to approximate as close as possible to the actual LD-50. The LD-50 of salt for radish seeds is around 40.199 g/L. This means that if a certain amount of seeds are put into a concentration with 40.199 g/L of salt, then 50% of the seeds would germinate while the remaining 50% would die. Conclusion:The data obtained from the LD-50 lab does not support the hypothesis "If the concentration of 18 g/L of salt is used on the radish seeds than half of the radish seeds will die because the radish seed will not be able to survive with high amounts of salt." In the hypothesis, it was predicted that 18 g/L of salt concentration would be the LD-50, after completing the lab, it can be concluded that the LD-50 is around 40.199 g/L. While the hypothesis was on the right track, that a higher concentration would be close to the LD-50, the prediction was not high enough. However, the data does support the idea that salt impedes and slows the growth of radish seeds. "Although, they need salt to perform their chemical procedure, too much of it can be fatal to terrestrial plants" (Arnold). An excess of salt has negative effects for plants. It can cause dehydration, which can cause the plants to wither and die, nutritional imbalance, and underdevelopment. The excess of salt on radishes seems to have caused them to be underdeveloped and unable to germinate. The higher amounts of salt could have also caused nutritional imbalances that would cause the seed to not be able to germinate.
Knowing how salt affects plants is important. Every year when the weather forecast predicts snow or ice, salt is put down on the roads in order to lower the freezing point of water. However, while this may keep people on the roads safer, it is actually harming the plants alongside the road. When the salt washes off the road and into the nearby plants, the salt impedes the plant's growth harming not only the individual plants, but the ecosystems that depend on those plants. There needs to be another way to prevent roads from freezing as much without harming the plants and ecosystems around them. Citations:
Tuthill,
Samantha-Rae. "Why Is Salt Used on Icy Roads?" AccuWeather.
AccuWeather, Inc., n.d. Web. 10 Feb. 2015. <http://www.accuweather.com/en/home-garden-articles/earth-you/icy-sidewalks-and-roadways-can/5631019>.
Arnold, Paul. "What Is the Effect of Saltwater on Plant Growth." Bright Hub. N.p., 13 June 2011. Web. 12 Feb. 2015. <http://www.brighthub.com/environment/science-environmental/articles/89127.aspx>. |
|